Extraction yield
The water/raw material ratio is a critical factor affecting extraction yield of active components from plant materials. Furthermore, the recovery of phenolic and flavonoid compounds is considerably influenced by polarity28. The purpose of the experiments in this study was to assess the effect of solvents on the extraction yield.
The results presented in Table 2 indicate that polar protic solvents had better extraction yields. Among all solvents used, ethanol and methanol achieved significantly higher extraction yields, with rates of 15.25â±â1.0% and 15â±â1.2%, respectively. This suggested that ethanol and methanol are optimal solvents for maximizing the extraction yield of F. communis fruit, followed by water, acetone, and ethyl acetate with yields of 7.9â±â0.25%, 2.6â±â0.05%, and 2.3â±â0.02%, respectively. However, hexane and chloroform achieved the lowest yields, with rates of 0.9â±â0.012% and 0.6â±â0.03%, respectively.
The polarity of the ethanol system is highly stable for the extraction of flavonoids and betacyanin glycosides, such as isorhamnetin-3-O-rutinoside, betanin, isobetanin, and betanidine, and polar components, such as phospholipids, polysaccharides, and lipoproteins20. Acetone and water are common extractants of proanthocyanidins and tannins12.
Our experiments show that the selection of an appropriate extraction solvent depends on the type and polarity of the constituents in the plant material.
HPLC analysis
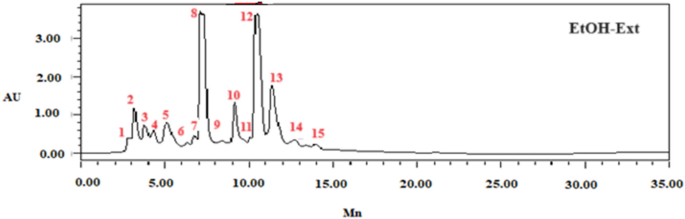
Fifteen phenolic compounds, including gallic acid, caffeic acid, catechin, 4-hydroxybenzoic acid, catechin hydrate, succinic acid, syringic acid, 3-hydroxybenzoic acid, naringin, cinnamic acid, ferulic acid, p-coumaric acid, sinapic acid, quercetin 3-O-β-d-glucoside, and rutin (Fig. 1), were identified and quantified using HPLCâDAD. In a previous study, various phenolic compounds, such as resorcinol, ferulic acid, syringic acid, and coumarin, are identified as the predominant components in F. communis fruits from various geographical origins. Compounds were identified according to relative retention times based on reference standards and reported as percentage areas (Table 3)7.
HPLCâDAD chromatogram of F. communis extract fruits (FC-Ext) at 320 nm using the following standards: Gallic acid (1), Caffeic acid (2), Catechin (3), 4-Hydroxybenzoic acid (4), Catechin hydrate (5), Succinic acid (6), Syringic acid (7), 3-hydroxybenzoic acid (8), Naringin (9), Cinnamic acid (10), Ferulic acid (11), p-Coumaric acid (12), Sinapic acid (13), Quercetin 3-O-β-d-glucoside (14), Rutin (15).
In a study focusing on F. communis rhizomes and employing a bioautography-guided isolation technique, three antibacterial sesquiterpenes were successfully isolated and characterized. These compounds were identified as daucane 14-(O-hydroxycinnamoyloxy)-dauc-4,8-diene, 2a-acetyl-6a-(benzoyl) jaeschkeanadiol, and 2a-acetyl-6a-(p-anisoyl) jaeschkeanadiol29. Additionally, ferulenol, a constituent of F. communis, has garnered attention owing to its various pharmacological effects, including antimicrobial, anticoagulant, antiproliferative, and antiappetitant activities. Recently, Gliszczynska et al., explored sesquiterpene coumarins, focusing on ferulenol, as promising lead compounds for drug discovery30. Notably, this diverse array of secondary metabolites substantially contributes to the antioxidant capabilities of F. communis while simultaneously conferring a broad spectrum of pharmacological and biological functionalities31. Phenolic acids, known for their various biological applications, are the primary polyphenols produced by plants and serve as precursors for bioactive molecules used in the therapeutic, cosmetic, and food industries. The health-promoting effects of these phenolic compounds are diverse and may include anticancer, anti-inflammatory, antioxidant, antimicrobial, and anti-aging properties32.
Total phenolic content
Phenolic compounds are the primary phytochemicals responsible for the biological activities of Ferula3. Moreover, the solvent influences the quantity and selectivity of the extracted components33. Phenolic compounds and other phytochemicals containing hydroxyl groups are primarily soluble in polar solvents34. As shown in Fig. 2a, our results present the effects of different extraction solvents (ethanol, methanol, water, hexane, ethyl acetate, and chloroform) on the total polyphenol content of F. communis. The ethanol extract exhibited the highest total polyphenol content (62.20â±â0.11 mg GAE/g DW), followed by the methanol (60.82â±â0.32 mg GAE/g DW) and aqueous (44.04â±â0.22 mg GAE/g DW) extracts. Acetone, ethyl acetate, chloroform, and hexane showed lower values at 19.76â±â1.19, 15.24â±â0.60, 13.94â±â0.92 and 6.93â±â0.43 mg GAE/g DW, respectively. Comparing the TPC of the solvents, the order established was: ethanolâ>âmethanolâ>âwaterâ>âacetoneâ>âethyl acetateâ>âchloroformâ>âhexane. The extract of Tunisian F. communis fruit exhibited the highest TPC (422 mg GAE/g DW), followed by flowers (207.21 mg GAE/g DW) and stems (129.86 mg GAE/g DW)35. In addition, Gamal and Atraimki quantified the total phenolics in n-butanol and ethyl acetate extracts (44.7â55.8 mg GAE/g DW)36.
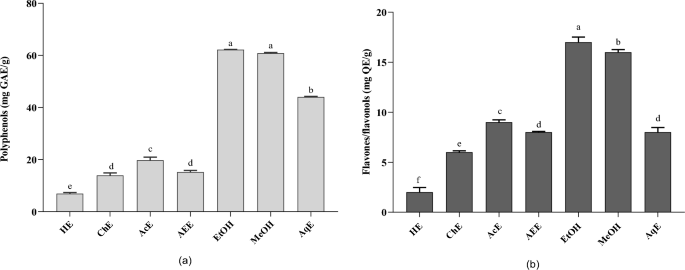
Effect of different solvent extractions on the (a) polyphenol content; (b) flavone/flavanol content; results sharing the same letter in the same test are not significantly different according to Tukeyâs multiple range test (pâ<â0.05). Data are the means of three replicates. HE, Hexane extract; ChE, chloroform extract; AEE, ethyl acetate extract; EtOH, ethanolic extract; MeOH, methanol extract; AqE, aqueous extract.
The extraction efficiencies varied considerably among the solvents, resulting in differences in the concentrations of total phenolics. The TPC of the extracts is influenced by the polarity of the extracting solvent and the solubility of chemical constituents in the solvent37. Solvents with greater polarity extracted more TPC29. Thus, more polar solvents such as ethanol, methanol, and acetone extracted more TPC. Ethanol extracts have the highest TPC, followed by methanol and acetone extracts17. This is consistent with a previous study, in which methanol and ethyl acetate extracted greater amounts of phenolic compounds than the other two37. These results confirm the critical role of extraction solvents in modifying the TPC recovery from plant material. Furthermore, based on the above results, it is clear that the ethanol and methanol extracts of wild F. communis fruit examined in this study exhibited comparable or greater efficiency of TPC extraction than previously reported.
Total flavonoid content
The total flavonoid content (TFC) from F. commnis exhibited a trend similar to that of TPC (Fig. 2b). The ethanol extract of Ferula exhibited the highest extraction yields of flavonoids (17.09 mg QE/g DW), followed by the methanol extract with 16.91â±â0.26 mg QE/g DW, and the acetone extract (9.31â±â0.24 mg GAE/g DW). Aqueous, ethyl acetate, chloroform, and hexane showed lower extraction yields with values of 9.31â±â0.24, 8.97â±â0.47, 8.68â±â0.08, 6.23â±â0.13, and 2.39â±â0.47 mg GAE/g DW, respectively. Owing to their polarities, the extraction of TFC significantly varied among the solvents. For the TFC, the extracts were classified in decreasing order of efficiency: ethanolâ>âmethanolâ>âacetoneâ>âwaterâ>âethyl acetateâ>âchloroformâ>âhexane. The methanol extracts of various aerial parts of F. communis L. showed the highest concentration in flowers, followed by fruits and stems, with values of 48.77, 14.23, and 13.37 mg QE/g DW, respectively. Our results, reflecting varying TFC amounts extracted by different solvents, can explain the presence of diverse groups of soluble flavonoids in F. communis fruits with distinct polarities. In addition, reported variations in bioactive compound values for Ferula in the literature may stem from factors such as varietal differences, environmental conditions, agronomic practices, and analytical methods.
Antioxidant activity of F. communis
Antioxidant activity was not assessed using a single antioxidant test30; rather, several in vitro procedures, including the DPPH, ABTS, RP, and TAC assays (Fig. 3), were used to assess antioxidant activity. As antioxidant test models vary in different respects, it is difficult to directly compare results9,34. Therefore, performing several screens using ex vivo models and in vivo studies is essential31. DPPH is a nitrogenous organic radical with free electrons, and is an easy and valuable spectrophotometric method for screening or measuring antioxidant activity38. When antioxidants react with DPPH, stable free radicals pair with hydrogen donors and are reduced to DPPH-H28. ABTS is a target molecule used to assess the reactivity of antioxidant samples in the presence of peroxides15. The radical cation of ABTS (ABTS+) is synthesized by oxidizing ABTS with potassium permanganate, potassium persulfate, or 2,2â²-azo-bis (2-amidinopropane)39. The ABTS assay reduces labor time, material cost, and sample volume, while decreasing color40. The RP assay is based on the principle that substances with a reducing potential react with potassium ferricyanide (Fe3+) to form potassium ferrocyanide (Fe2+)41, consequently reacting with ferric chloride to form a ferric-ferrous complex with an absorption maximum at 700 nm42.
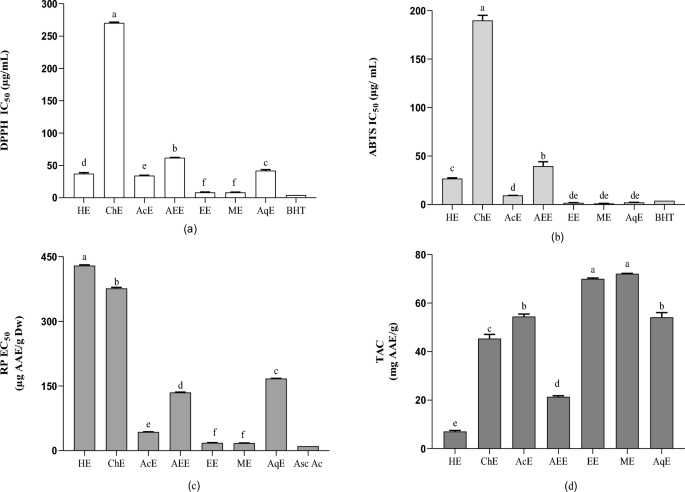
Antioxidant activities: (a) IC50 of the DPPH test; (b) IC50 of the ABTS test; (c) EC50 of reducing power; (d) the content of total antioxidant capacity (TAC). Each response is the average of triplicate with standard error. Values in the same column followed by different letters are significantly different according to Tukeyâs multiple range test (pâ<â0.05). HE, Hexane extract; ChE, chloroform extract; AEE, ethyl acetate extract; EtOH, ethanolic extract; MeOH, methanolic extract; AqE, aqueous extract; IC50, the extract concentration provides 50% inhibition; DPPH, 2,2âdiphenylâ1âpicrylhydrazyl; ABTS, 2,2â²-azino-bis (3-ethylbenzothiazoline-6-sulphonic acid); TAC, total antioxidant capacity.
The greatest DPPH scavenging capacity was demonstrated by the methanol extract with an IC50 value of 8.09 µg/mL, followed by the ethanol, acetone, and aqueous extracts with IC50 values of 8.25, 34.23, and 37.28 μg/mL, respectively. The scavenging efficacy decreased in the following order: ChEâ<âAEEâ<âAqEâ<âAcEâ<âEtOHâ<âMeOH. These results demonstrated that highly polar solvents, including ethanol and methanol, are effective for the extraction of antioxidants with efficient free radical scavenging properties and are more effective than intermediate-polarity solvents, such as acetone and hexane, but not chloroform (Fig. 3a).
The results of this study are consistent with those of previous studies wherein potent antioxidant activities were observed for both ethyl acetate (IC50â=â4.4â±â0.1 μg/mL) and n-butanol extracts36. The antioxidant activity assessed by measuring the scavenging power of DPPH on gum resin of the Algerian F. communis L. harvested in the Sahara had an IC50 of 5.08 mg/mL43. Roots of another species, Ferula gummosis Boiss, was evaluated for antioxidant activity using the DPPH assay and displayed an IC50â=â579.6â±â19.4 μg/mL44. The RP of F. communis extracts was highest in the MeOH extract, with an EC50 of 17.63 µg, followed by the ethanol, acetone, and ethyl acetate extracts, with EC50 values of 17.63, 43.4, and 135 µg AAE/g DW, respectively. Hexane exhibited the lowest RP with an EC50 of 429.3 μg AAE/g DW. The high RP of the F. communis methanol extract is probably due to clusters of hydroxyls in phenolic compounds, serving as electron donors. Therefore, antioxidants act as both reducers and activators45. The RP of a compound can considerably indicate its antioxidant potential46. However, no study has evaluated the RP of F. communis fruits.
The results of the ABTS assay showed significant variations among the extracts (Fig. 3b). The ethanol extract expressed the highest percentage of ABTS free radical inhibition (IC50â=â1.88â±â0.07 μg/mL). Alternatively, the chloroform extract (ChE) showed the lowest ABTS radical scavenging capacity (IC50â=â189.87 μg/mL). The activity (ABTS) results for F. communis extracts have not been previously published.
Reduced antioxidant activity, as determined by the TAC assay (Fig. 3d), was similar to the activities determined by the other tests. The MeOH and EtOH extracts exhibited the highest values (72.15â±â0.11 and 70.04â±â0.34 mg AAE/g, respectively). In addition, the hexane sample showed the least RP (7.05â±â0.46 g AAE/g). The methanol, ethanol, acetone, and aqueous extracts showed powerful and significant correlations in the four antioxidant tests (Table 4), possibly related to their phytochemical constituents, primarily phenolic components. These results supported the investigation of the antimicrobial activity of these extracts.
Antimicrobial studies
Antibacterial activity
The antimicrobial activities of F. communis extracts, including EtOH, AqE, and AcE, were evaluated against the examined microorganisms. EtOH and AcE extracts exhibited significant antibacterial activity compared to the reference antibiotic, oxacillin, as determined by the inhibition zone diameter and MIC values (Tables 5, Fig. 4). The AcE extract displayed the highest inhibitory activity against P. mirabilis with an inhibition diameter of 19.00â±â1.00 mm and an MIC of 2.50â±â0.00 mg/mL, followed by E. coli with 11.50â±â1.50 mm of inhibition zone and an MIC of 2.50â±â0.00 mg/mL. The extract also exhibited inhibitions of 11.00 and 9.00 mm against S. aureus and B. subtilis, respectively.

Visual observations of the inhibitory effects of Ferula communis extracts against pathogenic bacteria, including Staphylococcus aureus ATCC6633, Escherichia coli K12, Bacillus subtilis DSM6333, and Proteus mirabilis ATCC29906.
The EtOH extract, which contained the largest amounts of TPC and TFC, also exhibited the greatest TAC and was the most effective against all bacteria. The greatest activity against E. coli, with an inhibition diameter of 14.00â±â1.00 mm, an MIC of 0.312â±â0.00 mg/mL, and the least zone of 9.00â±â0.00 mm, was observed for S. aureus. AcE was more potent than EtOH for both extracts. Both extracts showed bactericidal activity against the four tested bacterial strains, whereas the aqueous extract of F. communis did not inhibit any of the examined strains. These differences in inhibition diameters can be attributed to the chemical composition of F. communis, and its antibacterial activity may primarily be because of the compounds in these extracts (EtOH, AqE, and AcE). The results showed that the ethanol and acetone extracts inhibited the four bacterial strains, with the ethanol extract having the best antibacterial activity.
Resorcinol, ferulic and syringic acids, and coumarin are renowned for their antibacterial activity. Compared to streptomycin sulfate, the antibacterial activities of the constituents extracted from the rhizome of F. communis revealed that the sesquiterpene 14-(hydroxycinnamoyloxy)-dauc-4,8-diene possesses vigorous activity against S. aureus, B. subtilis, E. faecalis, and S. durans7. The 80% ethanol-solubilized fraction of F. communis exhibited the greatest antibacterial effects on all assayed bacteria, whereas the aqueous-solubilized fractions did not exhibit any effect on the clinical isolates of Strep. pyogenes and Strep. pneumonia47. Alternatively, the isolated oil of Tunisian Ferula lutea was tested for its antimicrobial activity using disc diffusion and microdilution assays against six gram-positive and five gram-negative bacteria and eight species of the genus Candida48. Oil from flowers of F. lutea exhibited antibacterial and anticandidal activity, with an MIC of 39Â g/mL against S. aureus, S. epidermidis, and E. coli, and an MIC of 156Â g/mL against C. albicans. The type of solvent used considerably affected the antibacterial activity of extracts of Ferula inflorescence. These antibacterial compounds were marginally more abundant in gram-negative bacteria than in gram-positive bacteria. This result is likely due to the shape and composition of the cell walls of gram-positive bacteria, which have a thick layer of peptidoglycan with teichuronic acid and glycopolymers such as teichoic acids, making them less sensitive to the action of plant extracts49.
Antifungal activity
In in vitro disk diffusion assays, the antifungal activities of F. communis extract against C. albicans, A. niger, A. flavus, and F. oxysporum were compared to that of the fungicide fluconazole. AcE exhibited significant activity against F. oxysporum, with a percent inhibition of 20.6â±â1.4% and an MIC value of 5.0â±â0.0 mg/mL. Additionally, it showed a percentage of inhibition of 19.0â±â1.0% and an MIC value of 5 mg/mL against A. niger. The EtOH extract resulted in a small zone of inhibition of only 12.50â±â2.50 mm) against C. albicans with an MIC of 2.50 mg/mL (Table 6, Fig. 5). Additionally, the ethanol extract exhibited an inhibition percentage of 26.0â±â2.0% and an MIC of 2.5 mg/mL against A. niger. The percent inhibition of F. oxysporum was 13.3â±â2.6 with an MIC of 5 mg/mL. The aqueous F. communis extract demonstrated antifungal activities against C. albicans with a 13.00â±â1.00 mm of inhibition zone and an MIC of 5 mg/mL for MIC, as this extract has no effect against A. niger and F. oxysporum. However, the three F. communis extracts did not exhibit antifungal effects against A. flavus.
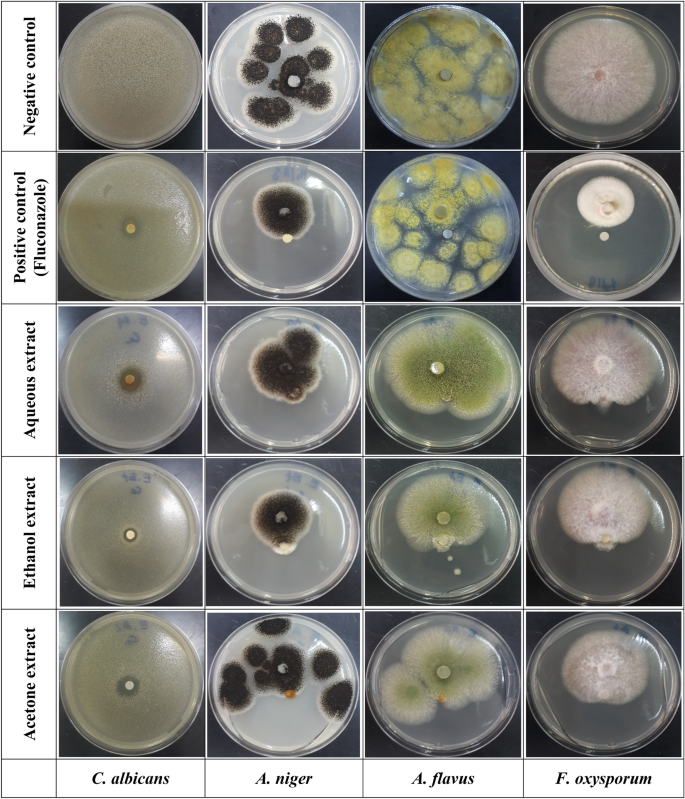
Visual observations of the inhibitory effects of Ferula communis extracts against pathogenic fungi, including Candida albicans ATCC10231, Aspergillus niger MTCC282, Aspergillus flavus MTCC9606, and Fusarium oxysporum MTCC9913.
Studies on the activity of F. communis extracts are scarce. More detailed information on the antimicrobial activities, especially the antifungal properties of F. communis, is required. The in vitro antibacterial activities of F. communis root extracts on colonies and conidia of Botryotinia fuckeliana, Penicillium digitatum, P. expansum, Monilinia laxa, M. fructigena, and Aspergillus spp were determined50; minimal antifungal activity on colony growth was observed, whereas root extracts exhibited the greatest effects on the growth of fungal colonies. However, the root extract was unable to inhibit conidia germination. These results are encouraging because the extract was not a pure product; therefore, the antimicrobial activity may be owing to several compounds related to the presence of bioactive metabolites.
Correlation
Multivariate analyses, such as principal component analysis (PCA), can reduce a complex dataset, make it more interpretable, and reveal relationships between or among parameters. In the present study, the correlations between antioxidant content, antioxidant activities, and antioxidant and antibacterial activities were examined. PCA results e(Fig. 6) showed that 91.2% of data variability is attributable to the first two principal components, PC1 (Component 1) and PC2 (Component 2), confirming that the correlation test is considered an excellent tool to reveal any relationship between different parameters studied. The first three PCs explained 100% of the data variance, which was sufficient to illustrate all the variables adequately. In this analysis, if two vectors subtend at a slight angle to each other, the two variables they represent strongly correlate. The plots of the scores for PC1 and PC2 showed a strong positive correlation among TPC, TFC, and TAC. The results of the DPPH, ABTS, and RP assays revealed a strong negative correlation between the bioactive content, as represented by the presence of phenolic and flavones/flavanol compounds, and antioxidant activity. Additionally, ethanol and methanol positively and negatively correlated with hexane, respectively. However, a positive correlation between DPPH and ABTS was observed, indicating that the most potent moieties for scavenging DPPH radicals showed the highest ABTS. The ingenious display of the polar heat map revealed a row and column hierarchical cluster structure in the data matrix (similarity/dissimilarity). This made it possible to group different samples based on their similarities. The analysis based on Pearsonâs correlation showed lower concentrations in brown and greater concentrations in dark blue (Fig. 7). Plant extracts of the genus Ferula have been arranged into two groups, one of which is the ethanol and aqueous extract. The polar heatmap also shows the correlation between the antioxidant content and antioxidant and antibacterial activities.
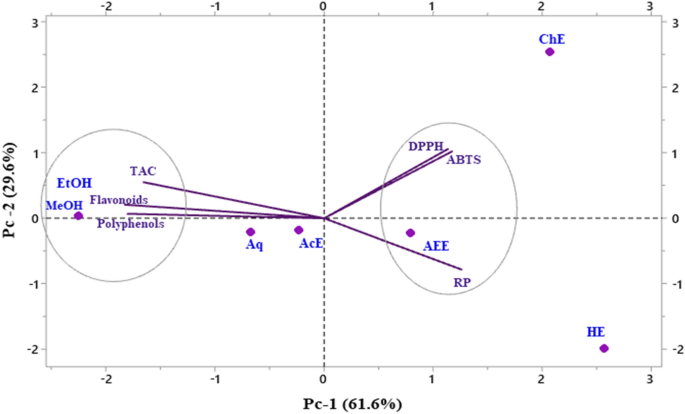
Principal component analysis.
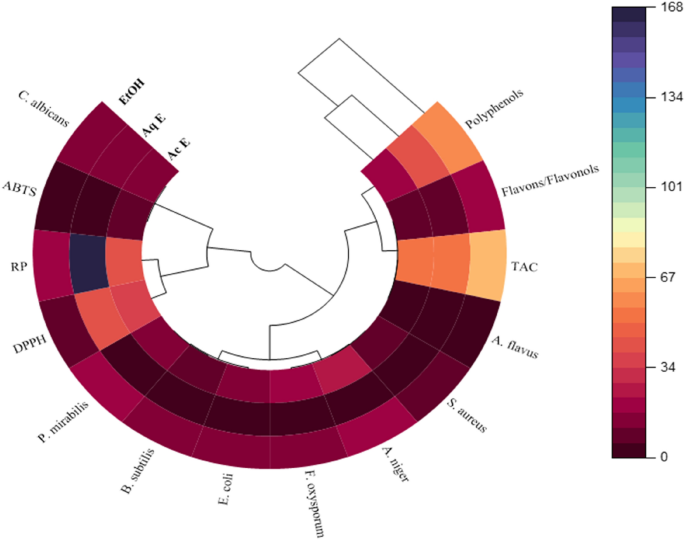
Polar heatmap with dendrogram for antioxidant and antibacterial correlations.
Molecular docking simulations
In investigating potential mechanisms of inhibition via antioxidant, antibacterial, and antifungal activities, nine molecular docking simulations were conducted on three targeted proteins with three major compounds of F. communis: syringic acid (C7), 3-hydroxybenzoic acid (C8), and p-coumaric acid (C12). These compounds were extracted at rates of 12.87, 17.34, and 29.42%, respectively (Fig. 8). Three major compounds were docked to the NADPH oxidase protein from Lactobacillus sanfranciscensis. Encoded by 2CDU.pdb, this protein exhibits binding energies of -5.28, -5.76, and 5.65 kcal/mol. It shares various common intermolecular interactions, forming hydrogen bonds with Ser115, Ala11, Lys134, and Gly12 amino acid residues (AAR) in the A chain. Additionally, it forms carbonâhydrogen bonds toward Thr9 AAR. When three major compounds were docked to the FimH lectin protein from E. coli K-12, encoded in the PDB by 4XO8.pdb, they formed another family of chemical bonds with binding energies of -5.38, -5.47, and -5.01 kcal/mol. Common bonds included one hydrogen bond with Arg92 AAR and one Pi-Pi alkyl bond with Pro91 AAR. This similarly was observed between syringic acid (C7) and p-coumaric acid (C12) (Fig. 9). 3-Hydroxybenzoic acid created another class of chemical bonds, forming six hydrogen bonds with Asp47, Asp54, Asp140, Phe1, Gln133, and Asn46 AAR, in addition to one Pi-alkyl bond with Il13 AAR in the A chain.
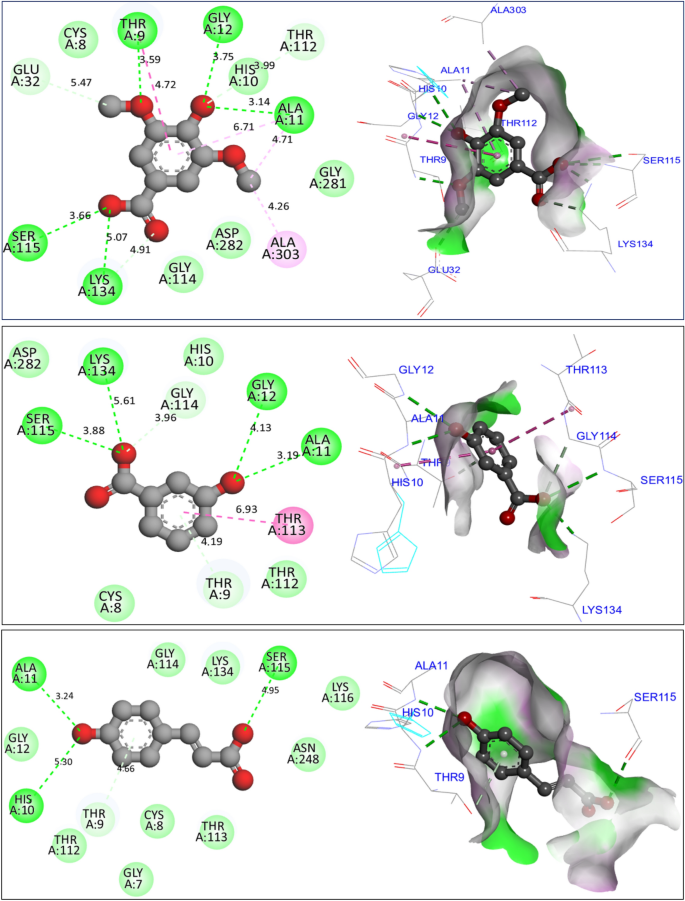
Results of intermolecular interactions between the NADPH oxidase protein from Lactobacillus sanfranciscensis (2CDU.pdb) and major compounds of Ferula communis (syringic acid, 3-hydroxybenzoic acid, and p-coumaric acid) in two and three dimensions.
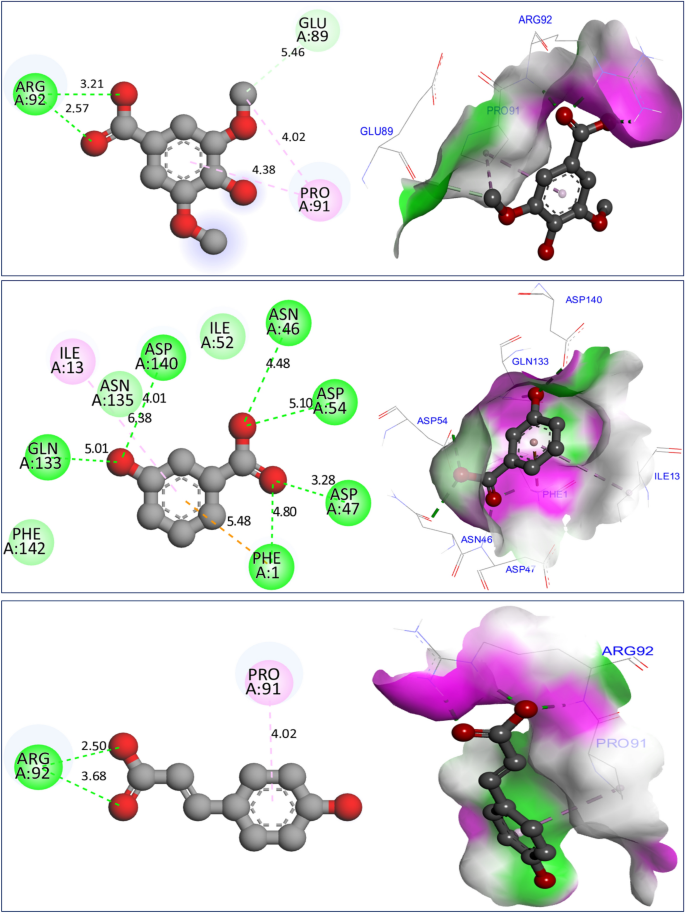
Results of intermolecular interactions between the FimH lectin protein from Escherichia coli K12 (4XO8.pdb) and major compounds of Ferula communis (syringic acid, 3-hydroxybenzoic acid, and p-coumaric acid) in two and three dimensions, produced.
The mechanism of antifungal activities of the three major constituents in F. communis extracts was examined by docking to the crystal structure of sterol 14-alpha demethylase (CYP51) from C. albicans encoded in the PDB by 5TZ1.pdb. The binding energies were -5.88, -6.93, and -6.77 kcal/mol, corresponding to C7, C8, and C12, respectively (Fig. 10). The major compounds of the studied plant produced similar chemical interactions, with three hydrogen bonds created with Arg381, Tyr118, and His468 AAR, and more than one bond resulting in a Cys470 AAR in the A chain of the targeted protein. The docking validation protocol for these nine molecular simulations was successfully evaluated. The three major compounds of the F. communis plant were strongly docked to the active sites of each targeted protein, as confirmed with the assistance of the protein plus server (https://proteins.plus/)51. The active sites of the proteins were defined as co-crystallized ligands (or native ligands) (Fig. 11). Three amino acids, Ala11, Lys134, and Thr9, were active sites for the NADPH oxidase protein. Amino acids Asp47, Asp54, Asp140, Phe1, and Gln133 AARs were the active sites of the antibacterial protein. Arg381, Tyr118, and His468 are the active sites of antifungal proteins. Binding energies in Kcal/mol are primarily negative and do not exceed the â 5.000 kcal/mol threshold, demonstrating molecular stability52. These results are consistent with those of Jeddi et al.53, who demonstrated antioxidant and antibacterial effects toward the same targeted receptors, NADPH oxidase and FimH lectin proteins, for two major compounds of the essential oil extracted from the mill of Lavandula angustifolia. In that study, the major compounds in F. communis extracts were similarly docked to the active sites of each responsible protein, sharing broadly equivalent intermolecular interactions. The biological activity of antifungal molecules from natural sources toward lanosterol 14-alpha demethylase (CYP51) involves similar intermolecular interactions, including the active sites of the CYP51 protein from C. albicans54.
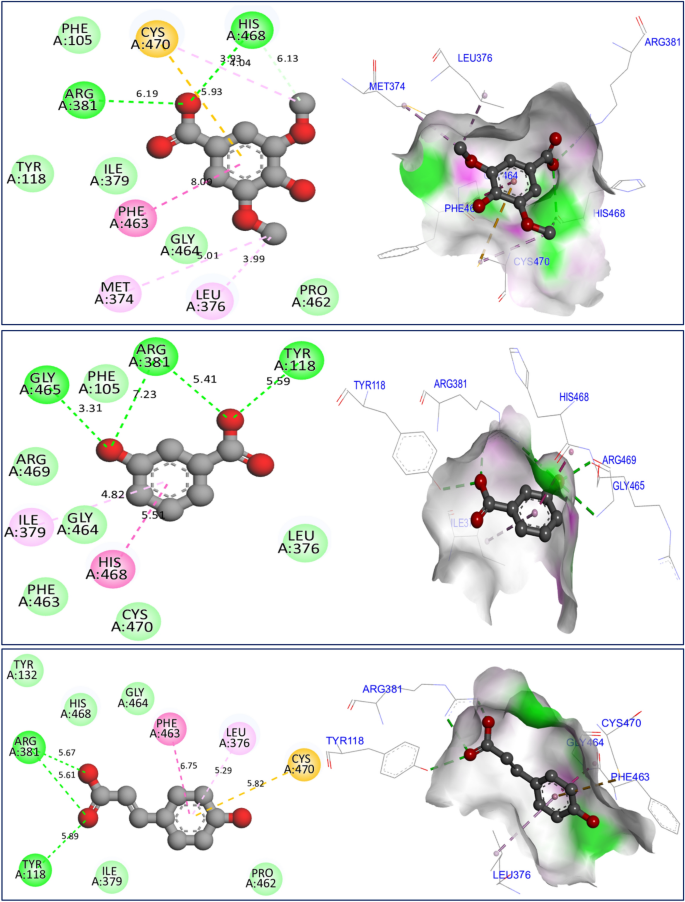
Results of intermolecular interactions between the CYP51 protein from Candida albicans (5TZ1.pdb) and major compounds (syringic acid, 3-hydroxybenzoic acid, and p-coumaric acid) in two and three dimensions.
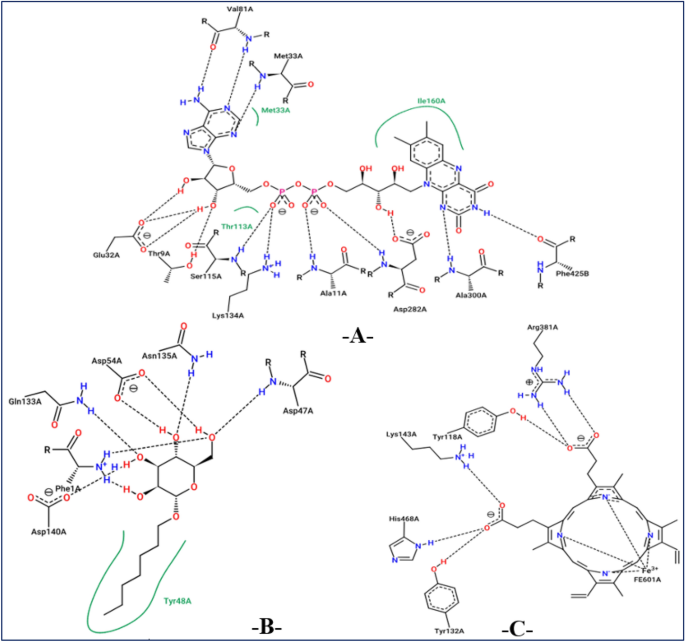
Active sites of antioxidant (2CDU.pdb), antibacterial (4XO8.pdb), and antifungal (5TZ1.pdb) proteins complexed with flavin-adenine dinucleotide (-A-), heptyl alpha-d-mannopyranoside (-B-), and tetrazole-based antifungal drug candidate VT1161 (VT1), respectively.