In the human body, molecules known as kinases propagate signals within and between cells, relaying signals that allow cells to respond to changes in the environment. However, there are hundreds of different kinases in the body, and identifying their individual and collective functions is challenging.
In a new study, Yale pharmacologist Benjamin Turk and his colleagues developed tools that can help researchers hone in on the roles of individual kinases and begin to uncover a more complete picture of their collective contribution to biological function.
And because dysfunctional kinases are often implicated in cancer, a more refined understanding of their function may yield better treatments in the future, they say.
The findings were published in Nature.
Kinases are enzymes that facilitate a process called phosphorylation. In cases that involve proteins, a protein kinase recruits a piece of a molecule called a phosphate group (a molecular fragment consisting of a phosphorus atom and four oxygen atoms) and helps attach it to a specific area of a protein known as a phosphorylation site. This can change the protein’s function in a number of ways, altering its activity or where it travels, for example.
There are two types of protein kinases depending on the proteins they phosphorylate: serine/threonine kinases, which Turk focused on in a previous study, and tyrosine kinases, the subject of the new study.
“Tyrosine kinases, in particular, are really important for cell-to-cell and organ-to-organ communication,” said Turk, associate professor of pharmacology at Yale School of Medicine. “The major class of tyrosine kinases is associated with growth factors. Understanding how tyrosine kinases signal is key to understanding how cells communicate with each other, with that communication often being a signal to grow or divide.”
All types of tyrosine kinases—of which there are 78 in humans—tend to over-send growth signals when they become hyperactivated, which is a key event in tumor growth, says Turk.
“This kind of study helps us understand the organization of tyrosine kinase signaling, which gives us insight into how kinases send growth signals and how blocking kinases might lead to a therapeutic response,” he said.
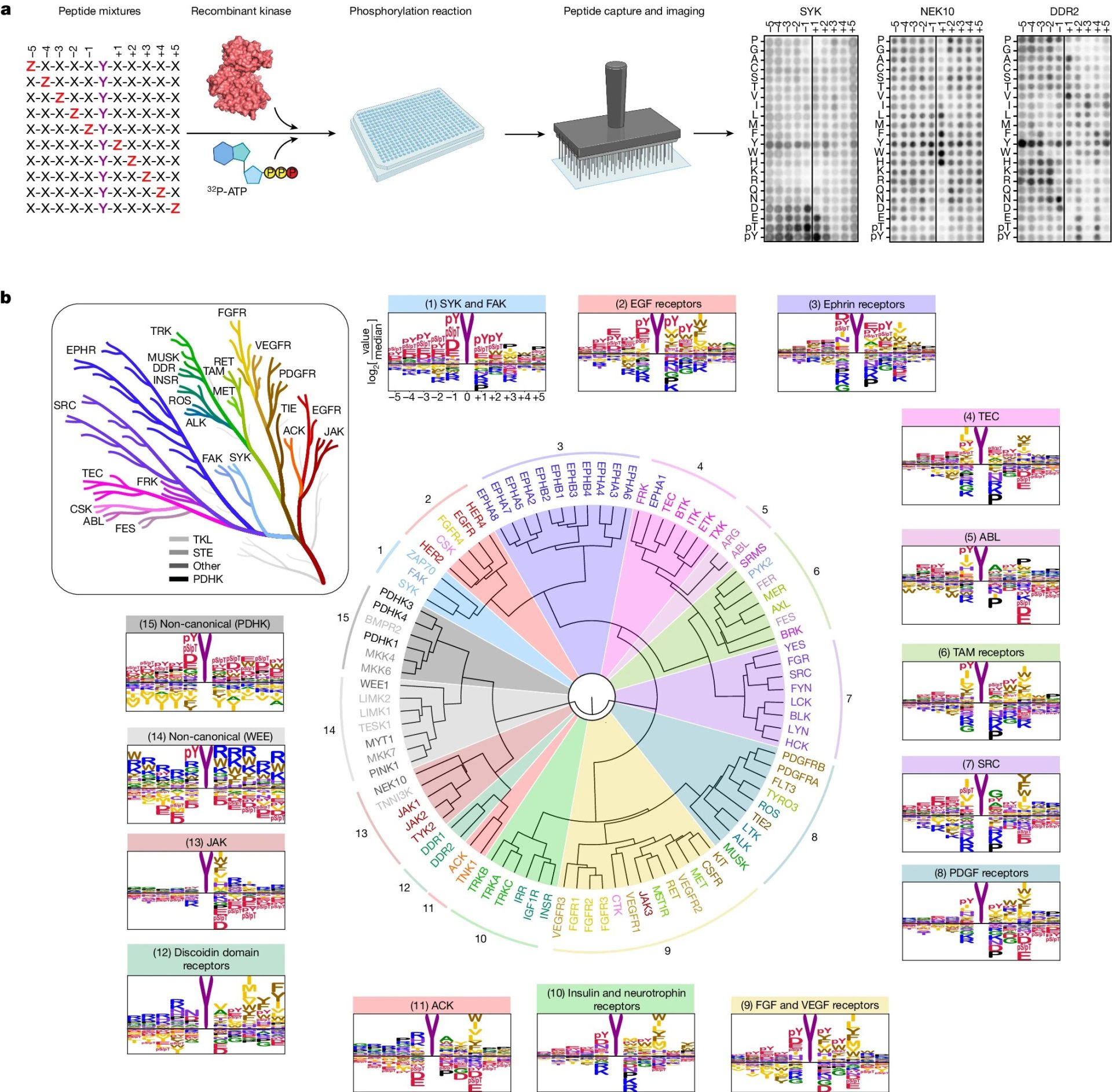
For the study, the researchers first looked at how kinases recognize their targets. Proteins are made up of amino acids, of which there are 20; kinases recognize short strings of amino acids that surround the site they phosphorylate.
Specifically, the researchers distributed each of the 78 tyrosine kinases into individual wells of laboratory plates, mixed them with a large number of different amino acid strings, and then looked at which strings the kinases preferred to phosphorylate. They then compared the kinases’ preferred strings to proteins in the human body.
“And we learned a few things by doing this,” said Turk.
First, they could, to some extent, start to match kinases to their targets in the body, which gives researchers information on the specific role of a particular kinase.
Maybe more importantly, their findings allowed them to uncover some of the broader rules of tyrosine kinase activity. It was as if they began to see the wiring of a house rather than just where individual outlets were, Turk said.
One of those rules has to do with how tyrosine kinases recruit additional kinases to propagate a signaling cascade. Another involves how amino acids surrounding a phosphorylation site dictate not just where phosphorylation will happen but also the rate at which it occurs.
And, importantly for treatment development, the tools developed in this study allow the researchers to infer which kinases might be active in a cell or tissue at a particular time and how perturbing them might affect their function.
“We can use drugs to inhibit individual kinases, and when we do that, we can see the activity of that kinase go down,” said Turk. “And tyrosine kinase inhibitors are one of the major targeted cancer treatments. But cancer cells can adapt to that kind of therapy and become resistant to it, causing patients to relapse.”
With their tools, the researchers can observe how blocking one kinase with an inhibitor sometimes leads to other kinases becoming overactivated, which may explain how cancer cells adapt and continue to grow. And that can help researchers develop more effective therapies, said Turk.
Going forward, Turk aims to use the rules uncovered in this work to start teasing apart key biological processes and to look further into how different cells respond to various kinase inhibitors.
But there’s another key takeaway of this work for him.
Tyrosine kinases are newer, evolutionarily speaking, than other kinases, emerging with multicellular organisms. When Turk and his colleagues compared human tyrosine kinases to those in nematodes—a type of worm that the human branch of the evolutionary tree diverged from millions of years ago—the specificity of both groups of kinases, or how particular they are about their targets, were extremely similar.
“That says this specificity—and understanding how it occurs—really matters,” said Turk. “It’s conserved throughout millions of years of evolution, and nature would not have maintained it in such fine detail without reason.”
More information:
Lewis Cantley, The intrinsic substrate specificity of the human tyrosine kinome, Nature (2024). DOI: 10.1038/s41586-024-07407-y. www.nature.com/articles/s41586-024-07407-y
Provided by
Yale University
Citation:
Getting to know the enzymes behind cell communication—and tumor growth (2024, May 8)
retrieved 13 May 2024
from https://phys.org/news/2024-05-enzymes-cell-communication-tumor-growth.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.