Cell lines
Cell line HEK293T was purchased from ATCC. Wild-type and CASP8-deficient Jurkat cells were generated by John Blenis (USA) and Clarissa von Haefen (Germany). Primary N4BP1+/+ and N4BP1â/â MEFs, kindly provided by Michael Kuehn (USA), were immortalized by transfection with plasmid containing SV40 T-antigen. All cells were regularly checked for Mycoplasma infection using VenorGeM Classic from Minerva Biolabs GmbH (Ltd).
Reagents
Puromycin and zeocin were from Invivogen. Ac-DEVD-cmk, z-IETD-fmk and z-VAD-fmk were from Santa Cruz Biotechnology. TNFα and IL-1β were from PeproTech. Polybrene and benzonase endonuclease were from Merck. Phosphatase and Protease Inhibitor Cocktails were from Roche Applied Science. Crystal violet was from Carl Roth GmbH and cycloheximide (CHX) from Enzo Life Sciences.
Plasmids and antibodies
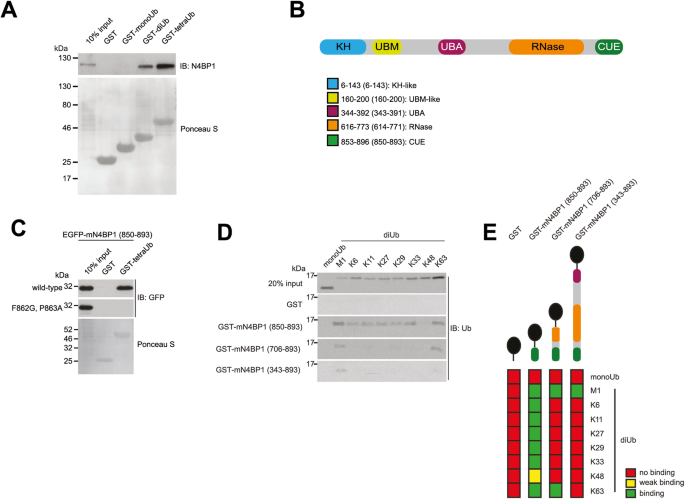
The lists of used plasmids and antibodies are available in Tables S1 and S2, respectively. In hexaUb YTH9 plasmid, five tandem Ub molecules had a terminal GG motif mutated to GV (to prevent cleavage by cellular DUBs during the screen), whereas the proximal Ub lacked GG motif (to prevent its potential conjugation to yeast proteins). For SPR experiments, the N4BP1 (613-774) and N4BP1 (613â893) fragments were cloned into pCold-TF (Takara) vector. For analytical gel filtration, the N4BP1 (613-774) and N4BP1 (613â893) fragments were cloned into pCold-I (Takara) vector, which include an N-terminal 20 amino acid solubility tag (GGGTPKAPNLEPPLPEEEKEG).
Yeast two-hybrid
Gal4BD-fused hexaUb plasmid was transformed into Y2HGold Saccharomyces cerevisiae strain (Clontech) and used as bait in Y2H screen, where it was mated with the human normalized cDNA library (Clontech) transformed into Y187 yeast strain (prey). Four independent reporter genes (AUR1-C, ADE2, HIS3, and MEL1) were used for selection according to the manufacturerâs instructions. Clone identities were determined by Sanger sequencing (Microsynth Seqlab).
Bioinformatics analysis
Multiple alignments were calculated using the L-Ins-I algorithm of the MAFFT package [53]. Sequence database searches were performed by the generalized profile method [54], using the pftools package. Inter-family similarities were established by Hidden Markov Model (HMM) to HMM comparisons, using the HHSEARCH programme package [55].
Transfection of mammalian cells
HEK293T cells were transiently transfected with either polyethylenimine (PEI, Polysciences) or GeneJuice transfection reagent (Merck Millipore). Non-adherent Jurkat cells were electroporated using the Neon Transfection System (Invitrogen). MEFs were transiently transfected using GenJet In Vitro DNA Transfection Reagent (SignaGen Laboratories). All the transfections were performed according to the manufacturerâs instructions. If the experiment required transfection of several plasmids simultaneously, appropriate amounts of empty vectors were used to equalize the total plasmid amount across the samples.
Retroviral production
For reconstitution of N4BP1â/â MEFs, various pBabe plasmids (carrying resistance to either puromycin or zeocin) were used. Twenty-four hours after seeding, HEK293T cells were transfected with appropriate plasmid DNA and helper plasmid phai by using GeneJuice. Thirty-six hours post-transfection, DMEM containing retroviruses was filtered, mixed with polybrene (final concentration 4âμg/ml) and transferred to target cells. Forty-eight hours post-infection, selection with either 300âμg/ml of zeocin or 4âμg/ml of puromycin was started. Cells were cultured in the presence of the appropriate antibiotics throughout use.
Generation of N4BP1 KO MEFs by CRISPR-Cas9
N4BP1 KOCRISPR MEFs were generated by transducing immortalized N4BP1+/+ MEFs with the lentiviral particles generated with the modified lentiCRISPRv2 (#52961, Addgene) single vector system [56]. Single gRNA against mouse N4BP1 (GAGTTGCAGCCAGATACGCG) was selected with the Azimuth 2.0 tool of the GPP sgRNA Designer and cloned into BsmBI site of the vector. Polyclonal N4BP1 KOCRISPR MEFs were selected with 2âµg/ml puromycin for 14 days and the obtained cell line was validated by Western blotting.
Various treatments of mammalian cells
Where indicated, starved cells were treated with either recombinant human IL-1β, mouse TNFα or human HS-TNFα at concentrations 10âng/ml, 20âng/ml and 1âμg/ml, respectively. To identify N4BP1 cleavage sites, serum-starved HEK293T cells were treated with recombinant mouse TNFα (20âng/ml) and additionally, with proteasome inhibitor MG132 (Tocris Bioscience) at 10 μM final concentration. For identification of the specific CASP responsible for N4BP1 proteolysis, MEF N4BP1+/+ and HEK293T cells were treated with recombinant mouse TNFα (20âng/ml) and 20âμM of either CASP inhibitors z-VAD-fmk, z-IETD-fmk or Ac-DEVD-cmk. For testing N4BP1 proteolysis in Jurkat cells, TNFα was used at concentration 100âng/ml. For cell death induction, recombinant mouse TNFα and cycloheximide (CHX) were used at concentration 10âng/ml and 0.5â1âμg/ml, respectively. For inhibition of apoptosis, cells were treated with mouse TNFα (10âng/ml), CHX (1âμg/ml) and z-VAD-fmk (20âμM). The duration of treatments is indicated in the figures.
Expression and purification of recombinant proteins
Various GST fusions were purified as shown previously [57]. HS-TNFα was expressed in E. coli with an N-terminal STREP II tag and purified on Strep-tactin Sepharose according to manufacturerâs instructions (GE Healthcare), followed by gel filtration using Superdex S75 (GE Healthcare) and subsequent dialysis against PBS. Recombinant active and inactive (C360S) CASP8 were purified from BL21 E. coli by lysing bacterial pellet in 50âmM Na2HPO4/NaH2PO4 (pH 7.4), 300âmM NaCl, 20âmM imidazole, 30% glycerol and 20âmM β-mercaptoethanol (β-Me). Precleared lysates were bound to nickel resins (Qiagen) and washed several times with lysis buffer, after which proteins were eluted with 50âmM Na2HPO4/NaH2PO4 (pH 8.0), 300âmM NaCl, 250âmM imidazole, 30% glycerol and 20âmM β-Me. N4BP1 proteins for SPR and ITC measurements were expressed and purified from BL21 E. coli using GST or immobilized cobalt affinity chromatography followed by size exclusion chromatography and anion exchange chromatography, if required. Size exclusion chromatography was performed in 50âmM HEPES (pH 7.4), 150âmM NaCl and 1âmM DTT, using Superdex S200 or Superdex S75 chromatography columns (GE Healthcare). For anion exchange chromatography, protein samples were separated by varied gradient elution from MonoQ columns (GE Healthcare) using 20âmM HEPES (pH 8.5), 1âmM DTT, 0.5% Triton X-100 and 0.1â1.0âM NaCl. For NMR spectroscopy 15N/13C-labelled N4BP1 (850â893) was expressed in M9 media (6âg/L Na2HPO4, 3âg/L KH2PO4, 0.5âg/L NaCl, 0.7âg/L 15NH4Cl, 2âg/L 13C-D-glucose, 10âmL/L 100X MEM vitamin solution (Gibco), 10 μM FeSO4, 10âμM CaCl2, 2âmM MgSO4, pH 7.4). For ITC and NMR experiments, the His-tag was removed from isolated N4BP1 CUE domain by incubation with 3âC protease at 4 °C, overnight. For analytical gel filtration, the His-tag of purified N4BP1 fragments was not removed. Enzymatic synthesis and purification of K48- and K63-linked Ub was essentially carried out as described [58].
Analytical size exclusion chromatography
Analytical size exclusion chromatography was performed using a SuperoseTM 12 10/300âL column (GE Healthcare) calibrated with the 29,000â700,000âDa GF Marker Kit (Sigma-Aldrich). Protein samples were eluted in 50âmM HEPES, pH 8.5, 150âmM NaCl, 1âmM DTT, 0.5% Triton X-100.
Isothermal titration calorimetry (ITC)
ITC experiments were performed at 293âK using a Microcal PEAQ-ITC calorimeter (Malvern). The protein solutions were prepared in a buffer containing 50âmM HEPES (pH 7.4), 50âmM NaCl and 0.5âmM TCEP. Experiments were performed at cell at concentrations 50â100âμM. The injectant concentration in the syringe was usually 10-fold to the titrant. For each titration 20 injections of 2âμl were performed. Integrated data, corrected for heats of dilution, were fitted using a nonlinear least-squares algorithm to obtain a binding curve, using the MicroCal Origin 7.0 software package. Each experiment was repeated at least twice, and average values are reported in Table 1.
In vitro CASP8 cleavage assay
C-terminally FLAG-tagged N4BP1 was transiently transfected into HEK293T cells, which were washed in 1xPBS 24âh after transfection. Cells were lysed in 1% Triton X-100 buffer (50âmM Tris-HCl, pH 7.5; 40âmM NaCl; 5âmM EDTA; 1% Triton X-100; 1x Protease inhibitor cocktail) and incubated on ice for 20âmin. Precleared lysates were incubated with M2 agarose (Sigma-Aldrich) at 4â°C for 2âh. After 5 washing steps with modified 1% Triton X-100 buffer (50âmM Tris-HCl, pH 7.5; 500âmM NaCl; 5âmM EDTA; 1% Triton X-100; 1x Protease inhibitor cocktail) and two washing steps with FLAG elution buffer (20âmM Tris-HCl, pH 7.5; 150âmM NaCl; 0.2âmM EDTA; 0.1% Triton X-100; 15% glycerol), FLAG-tagged N4BP1 was eluted from M2 resins with FLAG peptide at concentration 3âμg/ml. Thirty microliters of eluate were incubated with 1âμg of either active or inactive (C360S) 6xHIS-tagged CASP8 cleavage assay buffer (50âmM HEPES, pH 7.2; 50âmM NaCl; 10âmM EDTA; 5% glycerol; 10âmM DTT) at 37â°C for 2âh.
GST and MBP pull-down assays
Pull-down assays were performed as described in [59]. Since the size of FLAG-N4BP1 (613â893) protein was identical to the size of GST diUb, GST diUb beads were additionally cleaved with 1U thrombin (GE Healthcare) in 1x thrombin cleavage buffer (20âmM TrisCl, pH 8.4; 150âmM NaCl; 2.5âmM CaCl2; 1âmM DTT) at 25â°C for 4âh after GST PD wash.
Pull-down of TNFR1-SC
For isolation of TNFR1-SC complex, cells were stimulated in the presence or absence of HS-TNFα at concentration 1âμg/ml for 5âmin. Then, cells were washed twice with ice-cold 1xPBS, lysed in TNFR1-SC lysis/PD buffer (20âmM Tris-HCl, pH 7.5; 150âmM NaCl, 1% Triton X-100; 10% glycerol; 2âmM NEM, 1x Protease inhibitor cocktail, 1x Phosphatase inhibitor cocktail) and incubated at 4â°C for 30âmin. Lysates were cleared by centrifugation (15,000ârpm, 4â°C, 30âmin) and supernatants precleared with Superflow resin (IBA GmbH) at 4â°C for 30âmin with rotation. HS-TNFα (1 μg) was added to non-stimulated control and lysates were incubated with prewashed Strep-tactin XT resin (IBA GmbH) for 2âh at 4â°C. Next, samples were washed 7 times with TNFR1-SC lysis/PD buffer, re-suspended in 1xLDS buffer supplemented with β-Me and denatured at 70â°C for 10âmin.
Ubiquitin-binding assays
One microgram of monoUb and synthetic diUb chains (UbiQ Bio) were incubated with indicated GST protein fusions bound to Glutathione Sepharose 4B resin in incubation buffer (50âmM HEPES pH 7.5; 150âmM NaCl; 1âmM EDTA; 1âmM EGTA; 1% Triton X-100; 10% glycerol and 1âmM DTT) for 2âh at 4â°C. Next, samples were washed four times with incubation buffer prior to elution in 1xLDS buffer supplemented with β-Me.
Surface plasmon resonance (SPR)
The interactions of TF-N4BP1 (613â893) with monoUb and M1-, K48- and K63-linked diUb were analysed by SPR, using a Biacore S200 (GE Healthcare). Experiments were performed in HBS-P+ buffer (10âmM HEPES, pH 7.4, 150âmM NaCl, 0.05% Tween 20) at 25â°C. TF-N4BP1 (613â893) (10âμg/ml) was covalently immobilised on a CM5 chip using the amine-coupling kit (GE Healthcare) according to the manufacturerâs instructions. Mono- and diUb proteins were dialyzed into HBS-P+ buffer prior to the experiments. Experiments were run with a concentration series of mono- and diUb at 30âμl/min with 20âs association and 30âs dissociation phases. Association and dissociation kinetics were too fast to be resolved in these experiments. Data analysis was therefore performed by analysing the plateau levels. KD values were obtained from non-linear least-square fitting using a hyperbolic binding equation in GraphPad Prism 8.
Immunoprecipitation experiments
For HA IP, cells were washed twice in ice-cold 1x PBS, lysed in HA lysis/IP buffer (20âmM Tris-HCl, pH 7.5; 150âmM NaCl; 0.5% sodium deoxycholate; 1% NP-40; 2âmM NEM; 1âmM PMSF; 1x Protease inhibitor cocktail) and incubated with benzonase endonuclease (4â°C, 30âmin). Lysates were centrifuged (13,000ârpm, 4â°C, 15âmin), followed by Sepharose CL-4B (Sigma-Aldrich) preclearing at 4â°C for 30âmin. Then, lysates were incubated with prewashed monoclonal anti-HA agarose (clone HA-7, Sigma-Aldrich) for 3âh at 4â°C, with agitation. Samples were washed three times with denaturing buffer and twice with 1x PBS, re-suspended in 1xLDS buffer supplemented with β-Me and denatured at 70â°C for 10âmin.
The procedure for FLAG IP was described in in vitro CASP8 cleavage assay protocol. Linear Ub IP was performed as described previously [60].
For endogenous IP cells were washed twice with ice-cold 1x PBS and lysed in endogenous IP buffer (20âmM Tris-HCl, pH 7.5; 150âmM NaCl; 1% Triton X-100; 2âmM NEM; 1x Protease inhibitor cocktail, 1x Phosphatase inhibitor cocktail) on ice for 30âmin. Lysates were collected and centrifuged (13,000ârpm, 4â°C, 15âmin), followed by incubation with the antibody recognizing desired protein and protein A/G Sepharose (Sigma-Aldrich) at 4â°C for either 4âh or overnight. Then, samples were washed four times with an endogenous IP buffer, and eluted by incubation in a 1xLDS buffer supplemented with β-Me (70â°C, 10âmin).
Immunoblotting
Immunoblotting was performed as in ref. [59]. Proteins were first separated by SDS-PAGE and transferred to either 0.22âμm (Santa Cruz Biotechnology) or 0.45 μm nitrocellulose membrane (NitroBind, Maine Manufacturing) using Bio-Rad apparatus for wet blotting. Transfer was performed in 1x transfer buffer (25âmM Tris; 190âmM glycine; 20% methanol), at constant amperage of 200âmA for 2âh. Next, membranes were either stained with 0.5% (m/v) Ponceau solution at RT for 20âmin or directly blocked in either 5% BSA in TBS-T (20âmM Tris-HCl, pH 7.6; 150âmM NaCl; 0.1% Tween 20) or in 5% milk solution in TBS-T at RT for 1âh. Incubation with indicated primary antibodies was carried out either at 4°C (overnight) or at RT for 1â2âh. List of antibodies is available in the Table S2. Then, membranes were washed three times with TBS-T for 10âmin, incubated with appropriate HRP-conjugated secondary antibodies at RT for 1âh, followed by three washing steps in TBS-T and TBS (20âmM Tris-HCl, pH 7.6; 150âmM NaCl). After incubation of membranes in either Western Blotting Luminol reagent (Santa Cruz Biotechnology) or Lumigen TMA-6 (GE Healthcare), protein signals were detected with Super RX-N X-Ray films (FUJIFILM Corporation) by using CURIX 60 developing unit (AGFA).
For linear Ub IP, immunoblotting was performed as described previously [60]. Eluates containing immunoprecipitated linear polyUb-modified proteins were separated on 4â20% gradient PAGErTM Gold gels (Lonza), transferred onto 0.22âμm nitrocellulose membranes by wet blotting at 30âV for 2âh, blocked in 5% milk solution in PBS-T at RT for 1âh, followed by incubation with 1F11/3F5/Y102L IgG dissolved in 5% milk solution in PBS-T at RT for 1âh. Membranes were washed three times with PBS-T, incubated with secondary HRP-conjugated goat anti-human antibody (RT, 1âh), washed four times with PBS-T and visualized as described above. For linear Ub IPs (where endogenous levels of modified proteins were visualized), as well as for endogenous co-IPs, Clean-Blot IP Detection Reagent (Thermo Fisher Scientific) was used instead of secondary HRP-conjugated antibodies.
Preparation of IP samples for MS analysis
For identification of cleavage sites in N4BP1, cells were transfected with plasmids encoding either FLAG-N4BP1 or N4BP1-FLAG. Twenty-four hours later, cells were washed twice in ice-cold 1x PBS, followed by lysis in denaturing buffer (20âmM Tris-HCl, pH 7.5; 150âmM NaCl; 1âmM EDTA; 0.5% NP-40; 0.5% sodium deoxycholate; 0.5% SDS; 1âmM DTT; 2âmM NEM; 1x Protease inhibitor cocktail; 1x Phosphatase inhibitor cocktail). After benzonase treatment (4°C, 30âmin), lysates were cleared by centrifugation (13,000ârpm, 4â°C, 15âmin). Next, lysates were incubated with prewashed M2 resins (Sigma-Aldrich) for 5âh at 4â°C, with agitation. Samples were washed three times with denaturing buffer and twice with distilled water and re-suspended in 1xLDS buffer supplemented with β-Me and denatured at 70â°C for 10âmin.
Mass spectrometry analysis of N4BP1 cleavage
After elution and denaturation, samples were resolved by SDS-PAGE and gel lanes were cut into 7 slices, reduced with 200âμl of 10âmM DTT, alkylated with 200âμl of 55âmM chloroacetamide and digested with trypsin (final concentration 20âμg/ml) at 750ârpm, 37â°C, overnight. Peptides were bound to C18 StageTips and separated on EASY-nLC 1000 UHPLC (Thermo Fisher Scientific) connected to Q-Exactive HF Hybrid Quadrupole-Orbitrap (Thermo Fisher Scientific) mass spectrometer. For peptide separation, 15âcm and 75âμm ID PicoTip fused silica emitters (New Objective) were used. Emitters were self-made packed with ReproSil-Pur C18-AQ 3âμm resin (Dr. Maisch GmbH). Elution of the peptides from the column was performed using a linear gradient of 7â38% solvent B (80% acetonitrile in 0.1% formic acid) in 20âmin with subsequent increase up to 95% solvent B within 5âmin, followed by re-equilibration to 5% solvent B. Mass spectrometer was operated in positive ion mode and MS spectra were acquired with following settings: a maximal injection time of 20âms and a 60,000/15,000 resolution at 200âm/z. Up to 15 most intense ions were selected for collision induced dissociation (CID) fragmentation. Data analysis was performed by using the MaxQuant software suite (version 1.5.3.30) and the internal search engine Andromeda and searched against the Uniprot Homo sapiens (released 2016) database. For the identification of cleavage sites in N4BP1, semi-specific tryptic peptides were searched for. Oxidation (M) and acetylation (protein N-terminus) were searched as variable modifications, whereas Cys carbamidomethylation (C) was set as fixed modification. Initial precursor mass tolerance was set to 4.5 ppm and MS/MS mass tolerance to 0.5âDa. Peptide and protein FDR (false discovery rate) was defined to 1%.
Subcellular fractionation
The cellular fractionation was performed as in [59]. For subcellular fractionation of HEK293T cell line, cells were washed once with ice-cold 1x PBS and re-suspended in ice-cold Fractionation buffer A (10âmM HEPES, pH 7.8; 10âmM KCl; 0.1âmM EDTA; 0.5% Triton X-100; 1âmM DTT; 1âmM PMSF; 1x Protease inhibitor cocktail; 1x Phosphatase inhibitor cocktail). After 10âmin incubation on ice, cell suspension was centrifuged (2000 rpm, 4â°C, 5âmin). Supernatant was designated as cytoplasmic fraction. The remaining cell pellet was washed twice in ice-cold buffer A. Next, cell pellet was re-suspended in Fractionation buffer C (50âmM HEPES, pH 7.8; 420âmM KCl; 0.1âmM EDTA; 5âmM MgCl2; 10% glycerol; 1âmM DTT; 1âmM PMSF; 1x Protease inhibitor cocktail; 1x Phosphatase inhibitor cocktail) by passing several times through narrow-gauge syringe, followed by 30âmin incubation on ice and centrifugation (13,000ârpm, 4â°C, 15âmin). Supernatant was designated as nuclear fraction.
For subcellular fractionation of MEFs, cells were washed twice with ice-cold 1x PBS, collected and centrifuged (800ârpm, 4â°C, 5âmin). After centrifugation, supernatant was aspirated and cell pellet was gently re-suspended in Isotonic lysis buffer (10âmM Tris-HCl, pH 7.5; 300âmM sucrose; 2âmM MgCl2; 3âmM CaCl2; 1x Protease inhibitor cocktail; 1x Phosphatase inhibitor cocktail). After incubation on ice, cell suspension was centrifuged (800ârpm, 4â°C, 5âmin). Supernatant was discarded and pellet was re-suspended in Isotonic lysis buffer by passing several times through narrow-gauge needle. Then, cell suspension was again centrifuged (13,000ârpm, 4â°C, 20âmin). Supernatant was collected and designed as cytoplasmic fraction, while pellet was re-suspended in Extraction buffer (20âmM HEPES, pH 7.9; 420âmM NaCl; 0.2âmM EDTA; 25% glycerol; 1.5âmM MgCl2; 1âmM DTT; 1x Protease inhibitor cocktail; 1x Phosphatase inhibitor cocktail) by passing several times through narrow-gauge needle. Suspension was incubated on ice for 30âmin with occasional shaking. After centrifugation (13,000ârpm, 4°C, 20âmin), supernatant was designated as nuclear fraction.
Luciferase assay
The assay was performed as in [59]. Measurements were done by using either a Wallac Victor3 1420 Multilabel plate reader (Perkin Elmer) or Synergy H1 Hybrid Multi-mode reader (BIOTEK). All experiments were done at least in biological quadruplicate, where each biological replicate consisted of technical duplicate.
Real-time PCR
The quantitative real-time PCR was performed with SensiMix SYBR & Fluorescein kit (Bioline) in the iCycler iQ5 Real-Time PCR Detection System (Bio-Rad). GAPDH was used as an internal control. The Comparative Ct (Threshold Curve) method was used for the quantification of the amount of target, normalized to internal control. List of oligonucleotides is available in the Table S3. Experiment was performed in biological triplicates, where each biological replicate consisted of technical duplicates.
Immunofluorescence
Cells were seeded on glass coverslips and treated as indicated in the text. Cells were fixed by using 2% PFA, permeabilized with PBS containing 0.2% Triton X-100, blocked in 5% BSA in PBS solution at RT. Next, cells were incubated with appropriate antibodies. Coverslips were mounted with Mowiol containing DAPI, in order to visualize nuclei. Images were acquired with LEICA TCS SP8 confocal laser microscopy. For quantification, at least 200 cells were counted per condition.
Crystal violet assay
The assay was performed as in ref. [61]. Optical density at 570ânm (OD570) was measured by using Wallac Victor3 1420 Multilabel plate reader (Perkin Elmer). Experiment was performed in biological triplicates, where each biological replicate consisted of technical quadruplicates.
Luminescent cell viability assay
Cell viability was determined by using CellTiter-Glo Luminescent Cell Viability Assay kit (Promega GmbH) according to manufacturerâs instructions. Briefly, 96-well plate was equilibrated at RT for 30âmin, followed by addition of 100 μl of CellTiter-Glo Reagent provided by the manufacturer. Cell lysis was initiated by shaking a 96-well plate for 2âmin. To achieve signal stabilization, 96-well plate was additionally incubated at RT for 10âmin. Measurement was performed by using Synergy H1 Hybrid Multi-mode reader (BIOTEK). Experiments were performed in biological triplicates, where each biological replicate consisted of technical duplicates.
NMR spectroscopy
All NMR samples (300-500 μM) were prepared in 20âmM phosphate buffer (pH 7.0), 50âmM NaCl, 1âmM DTT, 10% D2O. For atom assignments, N4BP1 CUE domain was uniformly 15N,13C-labelled and assignments were completed using standard triple-resonance assignment methodology [62]. A total of 97% of the potential backbone (disregarding the proline residues) and 87% of the potential side-chain resonances were assigned (the first 3 N-terminal residues from the tag are ignored). Titration experiments involving 15N-labelled N4BP1 CUE domain were performed by addition of up to 5 molar equivalents of unlabelled Ub. Titration experiments involving 15N-labelled Ub were performed by addition of up to 5 molar equivalents of unlabelled N4BP1 CUE domain. The magnitude of chemical shift perturbations (CSPs) for each resonance was quantified according to the equation Î\({\rm{\delta }}\) = ((\({\rm{\delta }}\)H bound–\({\rm{\delta }}\)Hfree)2â+â((\({\rm{\delta }}\)N bound–\({\rm{\delta }}\)Nfree)/a)2)1/2, where a = (\({\rm{\delta }}\)Nmax– \({\rm{\delta }}\)Nmin)/ (\({\rm{\delta }}\)Hmax– \({\rm{\delta }}\)Hmin). NMR experiments were performed on two types of Bruker spectrometers, an AvanceNEO 600 equipped with a 5âmm 1H/13C/15N inverse triple resonance probe and an Avance III HD 700, equipped with a 5âmm 1H/13C/15N triple-resonance PFG cryoprobe. All spectra were collected at 303.15âK. Data were processed using NMRPipe [63] and analysed using CcpNmr Analysis V2 [64].
NMR structure determination
An experimentally guided model of N4BP1 CUE domain was generated with NMR chemical shift data in combination with homologous structural information using the standard CS-Rosetta method [65, 66]. Backbone chemical shift data (Cα, Cβ, Câ, N, Hα and HN) was included and a total of 20,000 models were generated. The top 10 models with the lowest energy were chosen as the final ensemble. Structural statistics were calculated using several servers including wwwPDB, MolProbity and PROSESS. Favourable Ramachandran statistics were observed, with 100% of residues in most favoured (98%) regions and 0% in outlier regions (Table S4).
Molecular modelling
The dimer of the C-terminal portion for the mouse sequence of N4BP1 was modelled with Robetta [67] (Comparative Modelling mode). The modelled region encompasses the RNase and CUE domains including their joining linker (residues 613â893 in the UniProt sequence Q6A037). The X-ray structure of the MCPIP1 dimer (PDB ID: 5H9W) was used as template for the dimer of the RNase domains (sequence identity = 52%), while the NMR structure from this work was used for CUE domain. Sequence alignments were generated using PRALINE [68]. Multiple models were generated, which showed high variability in the relative arrangement of the CUE domains with respect to each other and to the RNase domains. This was consistent with the partially disordered and thus highly flexible nature of the linker domain (residues 776-849) as predicted by DISOPRED3 [69]. Correspondingly, the estimates of model local error were generally low for the RNase domain (1.2âà on average) and high for the linker (> 20âà ). The best model was selected to have a distance between the centres of mass of the two CUE domains compatible with a simultaneous binding to M1-linked diUb (~ 31âà ).
A model of the CUE/monoUb interface was built using HADDOCK2.4 [70] with default parameters. The structure of monoUb was taken from PDB ID: 1UBQ. CSP values were used to define the Ambiguous Interaction Restraints for the calculation. In particular, residues with CSP values greater than the average value calculated over each molecule and with a relative solvent accessible surface area (SASA) larger than 30% were set as active residues. SASA values were calculated using GetArea [71]. The solution with the best HADDOCK score (Z-scoreâ=ââ1.3) was also the one most consistent with the interface model emerging from the experimental data from this work and in particular with the involvement of the nonpolar interface formed between the hydrophobic patch surrounding I44 of Ub and the FP motif of the CUE domain, as well as the polar contact between K48 of Ub and D893 of N4BP1.
The final model of the N4BP1 dimer bound to the M1-linked diUb was built with MODELLER 9.15 [72]. A template of the CUE/diUb complex was built by superimposing a copy of the CUE/monoUb best model from HADDOCK on each Ub molecule in the experimental structure of M1-linked diUb (PDB ID: 2W9N). The best Robetta structure (see above) was used as a template for the N4BP1 dimer. For each MODELLER run, 100 structures were generated and the one with the lowest DOPE score was selected as the final structure.
The resulting N4BP1/M1-linked diUb model was refined by energy minimisation using GROMACS 2016.3 [73]. The system was solvated using a truncated octahedral box of TIP3P water molecules. A minimal distance of 12âà was set between the protein and the walls of the box. The proteins were described with the Amber99SB*-ILDN [74] force field. The charge of the ionisable residues was set to that of their standard protonation state at pH 7, the systems were then neutralised by adding counter-ions. Each system was minimised through 3 stages with 7000 (positional restraints on heavy atoms) + 5000 steps of steepest descent, followed by 2000 steps of conjugate gradient. The quality of the refined models was evaluated using MolProbity [75]. The refined model had a MolProbity score <=1.62 (92nd percentile) and a clashscore <= 0.7 (99th percentile).
Statistical analysis
To determine statistical significance in Fig. S2E, an unpaired, two-tailed Studentâs t test was used. Three independent experimental replicates consisting of technical duplicates were performed. To determine statistical significance in Fig. S2C, a two-way ANOVA test was used. Five independent experimental replicates consisting of technical duplicates were performed. To determine statistical significance in Figs. 2E, 3A, 3C and S2F, a two-way ANOVA test, post hoc Sidakâs multiple comparisons test was used. Three independent experimental replicates consisting of technical duplicates were performed in Fig. 2E. Three independent experimental replicates consisting of technical triplicates were performed in Fig. 3C. Three independent experimental replicates consisting of technical quadruplicates were performed in Fig. 3A. To determine statistical significance in Fig. 3F, a two-way ANOVA test, post hoc Tukeyâs multiple comparisons test was used. Three independent experimental replicates consisting of technical duplicates were performed. For all of the figures, results are shown as means and error bars defined as s.e.m. ****Pâ<â0.0001, ***Pâ<â0.001, **Pâ<â0.01, *Pâ<â0.05 were considered significant, while Pâ>â0.05 was considered nonsignificant. No data were excluded for analysis.